Abstract
Introduction
Recent advances in the understanding of the WM pathobiology led to the expansion of our therapeutic options in this disease. A number of targeted drugs are either available or under investigation and are poised to change the chemo-immunotherapy paradigm in the treatment of WM. Now more than ever, an individualized treatment approach for patients with WM is possible, where patient characteristics and preferences can be matched by different treatment goals and side effect profiles. While deep responses have been associated with longer disease-free survival in other hematologic malignancies, data on patients with WM are sparse. We report the impact of depth of response in disease-free and overall survival (OS) in WM.
Methods
WM patients consecutively seen at Mayo Clinic between 01/1998 and 12/2016 were reviewed for response to therapy and disease burden. The best response achieved after the treatment of interest (TOI) was classified using the IWWM-7 Response Criteria. The included TOI were: BR, DRC, BDR and high-dose chemotherapy followed by ASCT. All time-to-event analyses were performed from the TOI initiation date using the Kaplan-Meier method and the log-rank test. A landmark analysis from the date-of-best response was also performed for OS to prevent immortal-time bias. Univariate and multivariate analyses of progression-free survival (PFS) and time-to-next therapy (TTNT) were performed using the Cox regression method.
Results
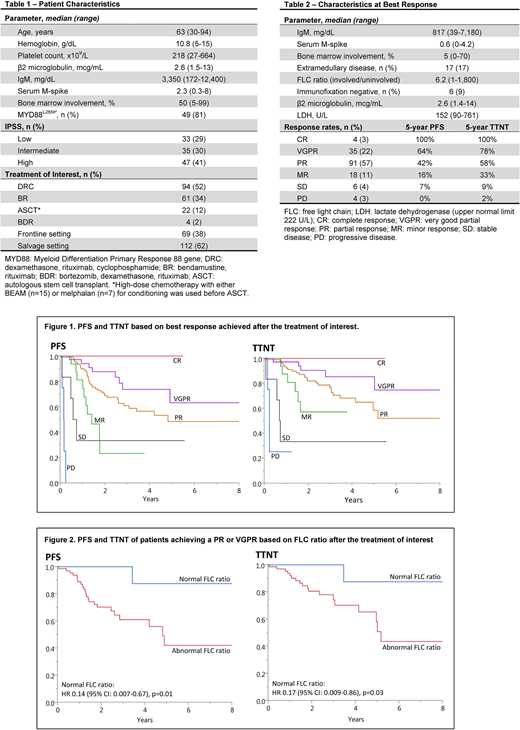
A total of 181 patients had disease response assessed after the TOI. Baseline characteristics at time of diagnosis are summarized in table 1. The median follow up from TOI was 3.8 years (95% CI: 3-4), the majority of patients received TOI as salvage therapy [n=112 (63%); median 2nd line (rage 1-11)]. Best response rates with the corresponding 5-year PFS and TTNT are summarized in table 2.
The median OS from TOI was longer in patients who achieved at least a PR [median 7.2 years (95% CI: 5-NR) vs 3.7 years (95% CI: 1.6-NR), p=0.01]. A trend towards a longer OS was also seen with deeper responses (PR or better) achieved with frontline therapy [median 5.8 years (95% CI: 5.8-NR) vs 3.4 years (95% CI: 1.6-NR), p=0.24]. Figure 1 shows the correlation between deeper responses with PFS and TTNT.
Among patients achieving a PR or VGPR, those with a normal FLC ratio post TOI demonstrated a longer PFS and TTNT [PFS: median NR (95% CI: 3.4-NR) vs 4.9 years (95% CI: 2.8-8.7), p=0.01; TTNT: median NR (95% CI: 3.5-NR) vs 5.2 years (95% CI: 4.2-9.4), p=0.04], figure 2. In a multivariate analysis including normal FLC ratio, normal IgM level and minimal BM involvement (<5%) post TOI, normal FLC ratio remained an independent prognostic factor for longer PFS and TTNT. Similar trends were seen when considering treatment naïve and relapsed/refractory patients independently (data not shown).
Discussion
Our results not only suggest an association between depth of response with PFS and TTNT, but also with OS in patients with WM. A normal FLC ratio (not part of the IWWM-7 response criteria) seems to predict a longer disease-free interval in patients achieving a PR or VGPR.
Gertz:Ionis: Honoraria; Apellis: Consultancy; annexon: Consultancy; Prothena: Honoraria; janssen: Consultancy; Physicians Education Resource: Consultancy; Alnylam: Honoraria; Teva: Consultancy; spectrum: Consultancy, Honoraria; Medscape: Consultancy; celgene: Consultancy; Research to Practice: Consultancy; Amgen: Consultancy; Abbvie: Consultancy. Kapoor:Takeda: Research Funding; Celgene: Research Funding. Ailawadhi:Amgen: Consultancy; Janssen: Consultancy; Takeda: Consultancy; Pharmacyclics: Research Funding; Celgene: Consultancy. Reeder:Affimed: Research Funding. Witzig:Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. Dispenzieri:Celgene, Takeda, Prothena, Jannsen, Pfizer, Alnylam, GSK: Research Funding. Lacy:Celgene: Research Funding. Kumar:Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; AbbVie: Membership on an entity's Board of Directors or advisory committees, Research Funding; KITE: Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding. Ansell:Seattle Genetics: Research Funding; Celldex: Research Funding; Bristol-Myers Squibb: Research Funding; LAM Therapeutics: Research Funding; Trillium: Research Funding; Pfizer: Research Funding; Takeda: Research Funding; Merck & Co: Research Funding; Affimed: Research Funding; Regeneron: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal